Electronic configuration of caesium12/4/2023 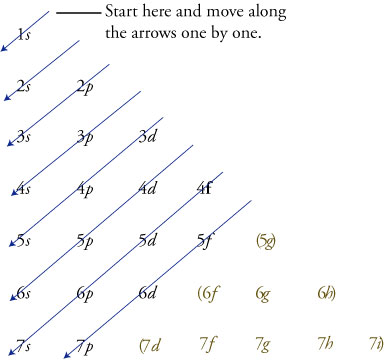
Germainum is in the 4th row Energy Level of the periodic table. The d orbitals Groups 3-12 (columns) can hold 10 electrons.Įach energy level must be filled before moving up an energy level.Įach orbital group must fill before moving to the next orbital group. The p orbitals Groups 13 - 18 (columns) can hold 6 electrons The s orbitals Groups 1 & 2 (columns) can hold 2 electrons The superscript tells us the number of electrons in the orbital. The Coefficient tells us the Energy Level (Row) of the periodic table The electron configuration for the first 10 elements 
The "f block" on the periodic table are the Lanthanide and Actinide series.Įlectron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The "d block" on the periodic table are groups 3-12 make up the d block and the elements' electron configurations end in d. The "p block" on the periodic table are groups 13-18 and end in p1, etc. The "s block" on the periodic table are groups 1 and 2 they end in s1 and s2. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons. The transition metals are behind by one period because the d electrons are high in energy.įor the rare earth elements (the Lanthanides and Actinides), they end in f. Scandium would end in 3d1, titanium in 3d2, etc. The general rule is that the element's electron configuration ends in d and whatever place they are in. And so it goes.įor the transition metals, groups 3-12, there are many exceptions. In group 4A or 14, all elements end in p2. Group 3A, or 13 all end their electron configurations in p1. Group 2 elements (2A), the alkaline earth metals, all end in s2 What period the element is in determines the 1st number.Įxample: H ends in 1s1 (even though H is not a metal, it resides in this group because it also has one valence electron) Group 1A (1), the alkali metals all end is s1. When looking at electron configuration, your fill order of electrons is: In essence, #"Ar"# is just a representation for #1s^(2)2s^2 2p^6 3s^2 3p^6# without us having to write it all out.Īll in all, the three given answers are correct ways of figuring out the ground-state electron configuration of Krypton. *Notice how the configuration #1s^(2)2s^2 2p^6 3s^2 3p^6# is the electron configuration for #"Ar"# so by starting with #"Ar"# we indicate that it is the configuration of everything that came before. The end result would then be: * # 4s^2 3d^10 4p^6# From there we write out the remaining configuration like we did in the first method. But we could also write out the configuration beginning using #"Ar"# since it is the closest noble gas with the lower energy. This method becomes really useful for elements that have a lot of electrons like Krypton because it becomes a hassle to write out such a long electron configuration.Ĭoincidently, Krypton itself is a noble gas so we could write the electron configuration as #. In essence, the shorthand notation tells us the configuration by using a noble gas element as our starting point instead of starting all the way at the #1s# orbital. The key to using this method is to identify the noble gas closest to the desired element that is at a lower energy (Has a lower atomic number if I'm loosely speaking). Using this as a guide and going through the periodic table we find the electron configuration to be: We know that the #s,p,d# orbital can hold a max of #2,6,10# electrons respectively. One way is to write out the entire electron configuration by going through each orbital or we can use a shorthand notation using the noble gases as a starting point. Third: Write out the electron configurationįor Krypton and most of the elements there are more the just one way (usually two) to write the electron configuration. (Refer to the following pictures as notes) Know how many electrons each orbital can hold and their order. First: Determine the number of electrons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
